Human Skeleton

WELCOME TO STRONTIUM FOR BONES BLOG
Have you experienced negative, and even dangerous, side effects from Fosamax (alendronate), Boniva (ibandronate), Actonel (risedronate), Reclast (zoledronic acid), Prolia (denosumab), Forteo (teriparatide), Tymlos (abaloparatide), or other drugs prescribed for osteoporosis? If you have, then rest assured there is a safe, effective treatment for this condition. Strontium, primarily in the form of strontium citrate, is taken orally once a day.
Visitors to my blog can leave comments or ask questions and can remain anonymous, if they wish. Their comments are relayed to my g-mail inbox. Below each post, the number of comments for that post is cited and underlined because it is a link. By clicking on that link below any post, a window opens so that a visitor can leave a comment. Ideally, visitors leave comments on posts most relevant to their comments. All comments to my posts are moderated by me.
Browse the posts and visit the link library of references.
Visitors to my blog can leave comments or ask questions and can remain anonymous, if they wish. Their comments are relayed to my g-mail inbox. Below each post, the number of comments for that post is cited and underlined because it is a link. By clicking on that link below any post, a window opens so that a visitor can leave a comment. Ideally, visitors leave comments on posts most relevant to their comments. All comments to my posts are moderated by me.
Browse the posts and visit the link library of references.
Saturday, January 25, 2014
Final Opinion on Possible Suspension of Strontium Ranelate to Be Made in February
The recommendation of the European Medicine Agency's Pharmacovigilance Risk Assessment Committee that strontium ranelate (Protelos and Osseor) should no longer be used to treat osteoporosis is still under consideration by the Committee for Medicinal Products for Human Use (CHMP). At its January 2014 meeting the Committee requested additional information from the company to inform its scientific decision making. A final opinion will be made by the CHMP at its meeting in February 2014.
http://www.nos.org.uk/
Labels:
CHMP,
final opinion,
Osseor,
Protelos,
suspension of strontium ranelate
Tuesday, January 14, 2014
What’s Behind the EMA’s Possible Suspension of Strontium Ranelate?
Here’s the official explanation:
"The EMA’s recommendations are based on an analysis of
pooled data from randomized studies in about 7,500 post-menopausal women with
osteoporosis. The results showed an increase in the risk of heart attack with
Protelos/Osseor as compared with placebo (1.7% versus 1.1 %), with a relative
risk of 1.6 (95% confidence interval, 1.07 to 2.38). There was also an
imbalance in the number of serious heart events seen with the medicine in two
other studies, one in men with osteoporosis and another in patients with osteoarthritis.
No increased risk in mortality was observed."
The increased risk of heart attacks did not show up in the
clinical trials of strontium ranelate, but it did show up in this analysis of
pooled dated from the trials. The results showed a relative risk of 1.6 for
heart attacks for the group on Protelos/Osseor compared to placebo. That result
means someone taking strontium ranelate has a 60 percent higher risk of heart
attacks than someone not taking the drug. That is quite significant.
Another reason the European Medicines Agency is considering
suspending strontium ranelate may be that the EMA is worried about the
possibility of experiencing with strontium ranelate what the French experienced
with Mediator, a drug manufactured by Servier, which also manufactures
strontium ranelate. The Mediator scandal became one of France’s biggest medical
scandals of recent years. French health experts now believe that Mediator, developed for
treating overweight diabetics, could have killed between 500 and 2,000 people between
1976 and November 2009, when it was finally banned for causing heart valve
problems and deaths. These numbers are disputed by Servier, which says there
are only three documented cases where death can be clearly attributed to
Mediator. In other cases, it says, aggravating factors were at work.
A final reason some believe is behind the EMA’s recommendations
and possible suspension is simply costs. Strontium ranelate is much more expensive
than alendronate (alendronic acid, generic Fosamax) for osteoporosis.
Sunday, January 12, 2014
Strontium Ranelate Prescriptions May Be Suspended in the European Union
The
European Medicines Agency's Pharmacovigilance Risk Assessment Committee (PRAC)
has recommended that strontium ranelate (Protelos/Osseor)
no longer be used to treat osteoporosis. The PRAC recommendation will now be
sent to the Agency's Committee for Medicinal Products for Human Use (CHMP), which
is expected to issue the Agency's final opinion at its meeting on January 20-23,
2014.
After further examination of the data, the PRAC noted that
there were four or more cases of both cardiac events and thromboembolism with
strontium ranelate for every 1000 patient-years compared with placebo. Moreover,
the drug is also associated with other adverse events, including serious skin
reactions, disturbances in consciousness, seizures, liver inflammation, and
reduced numbers of red blood cells.Although strontium ranelate does prevent about five nonspinal fractures, 15 spinal fractures, and 0.4 hip fractures for every 1000 patient-years, the committee decided that these benefits did not outweigh the potential risks and therefore the product's use should be suspended until further data support a benefit in a defined patient group.
In April 2013, the PRAC advised restrictions on use of the product, and those restrictions were endorsed by CHMP.
Servier, the manufacturer of strontium ranelate, acquired
the patent rights to market strontium malonate everywhere except in the United
States, where Osteologix retained the rights. Perhaps, now, Servier will be
more inclined to develop and market strontium malonate, which may not have the
problems associated with strontium ranelate. However, this is a possibility for
the future, not the present.
Strontium citrate, which also may not have the problems
associated with strontium ranelate, is currently an option for any patient in
the EU who wants to continue taking strontium, if strontium ranelate use is
suspended. I have been taking strontium
citrate continuously for six years with no problems.
Subscribe to:
Comments (Atom)
Wandering Skeleton

Artist: Joel Hoekstra
Osteoporotic Bone
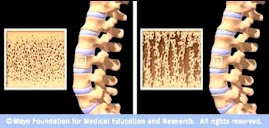
Source: www.mayoclinic.com
How Strontium Builds Bones
Strontium is a mineral that tends to accumulate in bone. Studies have shown that oral doses of strontium are a safe and effective way to prevent and reverse osteoporosis. Doses of 680 mg per day appear to be optimal. See my "For More Information About Strontium" links section.
Osteoporosis is caused by changes in bone production. In healthy young bones there is a constant cycle of new bone growth and bone removal. With age, more bone is removed and less new bone is produced. The bones become less dense and thus more fragile.
Scientists believe that strontium works in two ways. It may stimulate the replication of pre-osteoblasts, leading to an increase in osteoblasts (cells that build bone). Strontium also directly inhibits the activity of osteoclasts (cells that break down bone). The result is stronger bones.
When taking strontium, be sure to take 1200 mg calcium, 1000 IU vitamin D3, and 500 mg magnesium daily. It is best to take strontium late at night on an empty stomach. Calcium and strontium may compete with each other for absorption if taken together.
Osteoporosis is caused by changes in bone production. In healthy young bones there is a constant cycle of new bone growth and bone removal. With age, more bone is removed and less new bone is produced. The bones become less dense and thus more fragile.
Scientists believe that strontium works in two ways. It may stimulate the replication of pre-osteoblasts, leading to an increase in osteoblasts (cells that build bone). Strontium also directly inhibits the activity of osteoclasts (cells that break down bone). The result is stronger bones.
When taking strontium, be sure to take 1200 mg calcium, 1000 IU vitamin D3, and 500 mg magnesium daily. It is best to take strontium late at night on an empty stomach. Calcium and strontium may compete with each other for absorption if taken together.
For More Information about Strontium
- A Dose-response Study With Strontium Malonate
- A Review of the latest insights into the mechanism of action of strontium in bone
- Antifracture Efficacy Over 10 Years With Strontium Ranelate
- Combination of Micronutrients for Bone (COMB) Study: Bone Density after Micronutrient Intervention
- Echolight REMS Scan of Young, Normal Female
- Effect of bone strontium on BMD measurements
- Effect of Lumbar Scoliosis on DXA Results
- Effects of SrR on Calcium Metabolism
- Effects of strontium ions on growth and dissolution of hydroxyapatite and on bone mineral detection
- Influence of strontium on bone mineral density and bone mineral content measurements by dual X-ray absorptiometry
- Interpretation of BMD Scans in Patients Stopping Strontium
- Melatonin-micronutrients Osteopenia Treatment Study (MOTS)
- National Osteoporosis Foundation
- Osteoporosis And Bone Physiology
- Post-Marketing Assessment of the Safety of Strontium Ranelate
- PubMed Abstract On The SOTI Study
- PubMed Abstract On The TROPOS Study
- Strontium ranelate Aristo
- Strontium Ranelate For Spinal Osteoarthritis
- Strontium: Breakthrough Against Osteoporosis
- Summary Safety Review - Strontium
- The Effects of Strontium Ranelate on the Risk of Vertebral Fracture in Women with Postmenopausal Osteoporosis
- The Influence of Strontium on Bone Tissue Metabolism and Its Application in Osteoporosis Treatment
- Thirteen Key Diagnostic Tests